MedTech Venture Builder Collaborative Centre of Excellence
Applications now closed – Starting July 2026
Applications now closed – Starting July 2026
The LIHE MedTech Venture Builder (MVB), now in its third year, is a 12-month structured programme in two stages to speed-up the commercial and clinical translation of promising medical technologies into robust, investment-ready healthcare ventures.
Supporting companies from across the UK, the MVB, the first of its kind, is a collaborative effort led by LIHE, in collaboration with leading NHS Trusts, industrial partners, prominent charities, and select impact investors.
Our collaborative focus on MedTech allows us to take ventures a significant step further than what has been achieved before programmatically. Understanding how to execute in this highly regulated and evidence-driven environment is key to attracting further dilutive and non-dilutive investment.
We provide the structures that are critical to de-risking your company for such investment, such as a clinical evidence roadmap and stakeholder mapping, a regulatory plan, extensive hands-on guidance and monthly strategic progress review, all while aligning with the requirements of a Quality Management System (QMS).
This is a truly unique opportunity for very select, high-potential MedTech companies from across the UK to receive fully customised, extensive executive support from experts in commercial and clinical translation from the partners.
The MVB was created with the support of a significant grant by Research England for a Collaborative Centre of Excellence (CCoE) for MedTech translation. The programme is further supported by our strategic partners, Team Consulting.



The MedTech Venture Builder programme is structured in two stages: Foundation and Execution, both focusing on key translational elements but at different depths.
The Foundation stage is free to join for eligible participants.
At the end of Foundation, select companies will be invited to join the Execution stage, where they will receive one-to-one, bespoke support. Terms and conditions apply (please read Programme information below) for joining the Execution stage.
You will develop a clear understanding of the interactions between all critical components needed to inform your strategic decisions, anchored around the evidence you need for clinical adoption.
You will develop an understanding of the regulatory landscape in key markets for your specific technology, with consideration of reimbursement models and adoption barriers.
You will develop an understanding of the governance rigour that you require to navigate clinical study approvals, including ethics, sponsorship and regulatory considerations.
Funding bodies look for cohesive translation plans they can fund. By combining the above elements into a coherent narrative, you will be in a better position to attract funding to accelerate your clinical validation.
You will develop an understanding of the mechanics of raising, and an enriched narrative from the learnings of the programme.
We work with you to build a clear clinical validation plan for your product, aligned to UK regulations, so you know what evidence you need, how to generate it, and what your first real clinical studies could look like for adoption of your technology.
We work with you to build a clear regulatory and go-to-market strategy for the UK and key markets for your technology, including investment needed.
We work with you to establish when in your roadmap is the best time for you to enable and initiate clinical studies and the best way to do so, with approximate costings.
We work with you to optimise your clinical and translation plans to inform funding decisions against your milestones, including identifying the most suitable sources of funding for your next steps and when best to pursue them.
Investor readiness at LIHE means being ready for due diligence. You’ll be mentored by an investor to confidently navigate meaningful conversations, backed by a validated go-to-market strategy, robust clinical plans and regulatory clarity.


By participating in the MedTech Venture Builder your venture will:
The MedTech Venture Builder invites applications from:
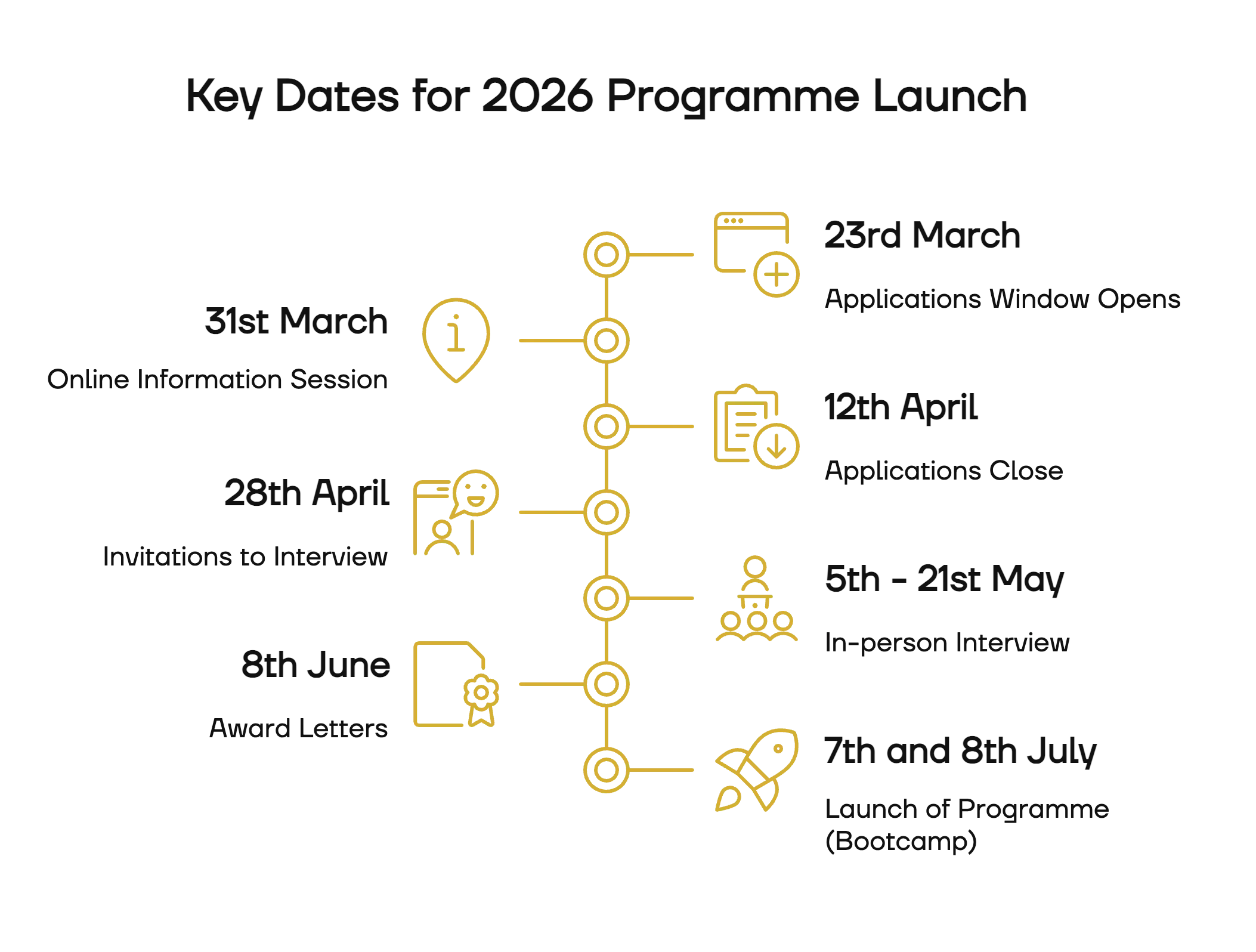
An applicant information session was held on 31 March for the MedTech Venture Builder. Watch the session below to find out more about the programme.

LIHE is the latest trailblazing initiative from King’s College London


